  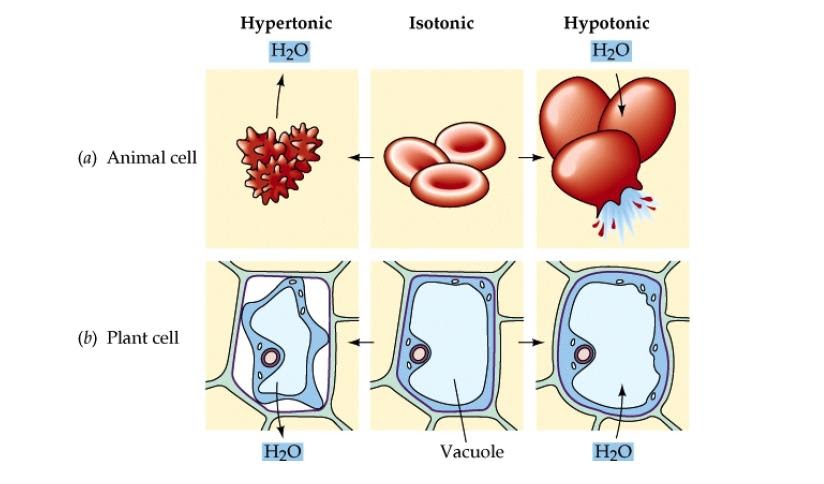
It is important to be familiar with what fluids are isotonic and when they are given. The cell has the same concentration on the inside and outside which in normal conditions the cell’s intracellular and extracellular are both isotonic. Easy Overview of Isotonic, Hypotonic, & Hypertonic Solutions Isotonic Remember when we are talking about isotonic and hypo/hypertonic we are talking about how it looks outside of the cell compared to inside. The key thing to remember here is that everything will move from a LOW concentration to a HIGH concentration. Osmosis allows molecules of the solvent to pass through a semipermeable membrane from a less concentrated solution to a higher concentrated solution. The cell loves to be in an isotonic state and when something happens to make it unequal (like with hypotonic or hypertonic conditions) it will use osmosis to try to equal it out. Each part is made up of a solution and depending on the tonicity of the fluid you can having shifting of fluids from outside of the cell to the inside via osmosis. The cell is divided into two parts: ( intracellular & extracellular). Isotonic, Hypotonic, & Hypertonic Fluids for Nursing Studentsįirst, let’s get familiar with the cell and how tonicity works through osmosis. Video Teaching Tutorial on Isotonic, Hypotonic & Hypertonic Solutionsĭon’t forget to subscribe to my YouTube channel for more teaching tutorials. In addition, I have an isotonic, hypotonic, and hypertonic quiz you can take to test your knowledge on this content. In this article, I give you an easy overview of each solution and how they work on the cellular level. In nursing school and on the NCLEX exam, you will be required to know what type of IV fluids are considered isotonic, hypotonic, and hypertonic. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
